From Biomedical Valley to Shenzhen, Lifemotion’s strategy focuses on clinical simplicity, modularity and regulatory pathway in all markets

Michael Van Driel pictured at his desk in Lifemotion’s headquarters, in a work context that recalls the link between technological development, business organization, and responsibility towards the clinical contexts of use, where design choices translate into operational solutions for the daily practice of extracorporeal therapy (Photo: Alice Luppi/Lifemotion)
In the global debate on the transformation of intensive care systems, the ECMO It is gradually moving beyond the realm of highly specialized technology to become a mobile clinical infrastructure, designed to integrate into emergency, transport, and continuity of care pathways. The evolution of life-support devices is not just about technological performance, but also impacts hospital organizational models, staff training, and the ability of healthcare systems to adopt complex solutions sustainably.
In this space he works Michael van Driel, Vice President of Business Development and General Manager of Lifemotion, a MedTech company active in the development of systems Extra Corporeal Life Support designed from the outset for long-term support and for use in highly variable clinical settings. His professional career, built between United States ed Europe, has been intertwined over the last two decades with the ecosystem of the biomedical district of Mirandola of Emilia-Romagna Region, where he contributed to the birth and industrial positioning of the Italian reality of the Chinese group from Shenzhen, integrating engineering skills, certified production chains and structured dialogue with clinicians.
The international experience of the Californian manager gained between American entrepreneurship, European regulation e Asian manufacturing capacity Lifemotion has helped define an approach geared toward the industrial scalability of intensive care technologies. Lifemotion operates along a value chain that combines design, manufacturing, and clinical validation, with increasing oversight of regulatory processes in Europe and a path to market access beyond Europe that requires alignment between regulatory requirements, reimbursement models, and economic sustainability for hospitals.
The development trajectory of the ECMO Today, innovation is also about reducing the operational complexity of devices, encouraging adoption in resource-limited settings, and enabling new use scenarios, such as extracorporeal support during transport or in out-of-hospital settings. From this perspective, co-design with physicians, nurses, and perfusionists becomes a structural element of innovation processes, as is the integration of monitoring systems aimed at early prevention of complications.
In a long chat, also supported by experience gained over the decades in other companies in the area such as Medtronic, TransMedics, LivaNova e Sorin Group, Michael van Driel analyses the main technological, clinical and regulatory challenges that are reshaping the field of Extra Corporeal Membrane Oxygenation, the role of industrial districts and global networks of expertise in building scalable solutions and the development directions that could, in the coming years, extend access to advanced life support well beyond the traditional confines of intensive care.

View of the entrance to the Lifemotion headquarters in San Giacomo Roncole, where engineering research, production, and corporate coordination intertwine with the biomedical ecosystem of the Mirandola district, a historic hub of expertise in the medical devices sector (Photo: Alice Luppi/Lifemotion)

Portrait of Michael Van Driel next to the ECMO machine, focusing on his torso and the system’s control interface. This image illustrates the operational dimension of the dialogue between management and biomedical technology in the development of devices intended for highly complex clinical contexts. (Photo: Alice Luppi/Lifemotion)
Throughout your career, you’ve said you directly contributed to the creation and development of Lifemotion in Emilia-Romagna. Can you clarify your specific role in the company’s launch and how the project’s initial governance was structured?
“I think I’ve been very fortunate because I’ve been able to learn so much from extremely knowledgeable people in the Mirandola area and the Emilia-Romagna region. Over the past twenty-five years, I’ve built relationships and business partnerships that have become friendships thanks to the common thread of the biomedical sector, particularly cardiac surgery. It was precisely these relationships and this expertise that made the launch of this company possible.”
How does the Lifemotion system differ from traditional ECMO devices in terms of design, clinical usability, and impact on workflows in intensive care units?
The Lifemotion system is unique in that it is a system specifically designed for long-term support, not a system originally designed for extracorporeal circulation and then converted to long-term support. Although the technologies are similar in construction, they are completely different in actual clinical use. Patients undergoing long-term support require systems designed to last, be transportable, and, most importantly, to meet the needs of those in critical conditions. This scenario is completely different from what is seen during a short-term intervention such as cardiac surgery.
Lifemotion aims to make extracorporeal membrane oxygenation accessible even in settings with limited resources. What are the main technological, educational, or healthcare system barriers, and how are you addressing them?
Extracorporeal membrane oxygenation therapy is inherently extremely complex and risky. The Lifemotion device was designed to be the simplest, easiest, and most intuitive system on the market. Long-term ECMO support is a very complex therapy to manage, and we tried to take this into account when designing the system. We even involved external experts in Europe and the United States to ensure the system was immediately adaptable for clinical use, so that resources and intensive care staff could focus on the patient and not on learning how to use the device. Ease of use and adaptability to existing clinical protocols were key to the system from day one.
Can you tell us how your over twenty years of international experience in the MedTech sector has influenced your strategic approach to leadership and innovation?
This has happened in many ways… Thanks to local technological developments in disposable equipment and devices, we have been able to translate this know-how into the quality of the products we manufacture, as well as the durability and consistency of their performance. The enormous progress made in the design and manufacturing of biomedical equipment has allowed products that twenty years ago were primarily identified as European to become international. The Lifemotion company has combined European know-how with the manufacturing power of a technologically advanced region like Shenzhen, China.
At the heart of many MedTech innovations is co-design with clinicians. How do you involve physicians, nurses, and perfusionists in the development and refinement of your products?
The ‘voice of the customer’ aspect is well regulated by ISO standards, Quality Management Systems, and the European Union’s MDR, but capturing the customer’s voice must be more than that. Our organizational culture allows us to react quickly to user requests, to constantly improve and simplify system use, and to drive innovation based on their needs. Patient monitoring is shifting toward the early identification of certain complications resulting from ECMO. Thanks to the contribution of physicians around the world, we are already taking measures to identify the early onset of infectious problems or inadequate support before these complications become irreversible.
Looking ahead to the next five to ten years, what technological or regulatory developments do you foresee in the ECMO field, and how does Lifemotion intend to position itself for that future?
“What we certainly envision for the next decade is faster and broader access. We can imagine that in the not-too-distant future, this type of life support could be made more available not only in emergency rooms, but also in ambulances. Making the technology and therapy rapidly deployable and easy to transport is the direction we will move in. We also want to integrate a range of cannulas into our product portfolio to meet the needs of the Extra Corporeal Membrane Oxygenation market as we know it today.”
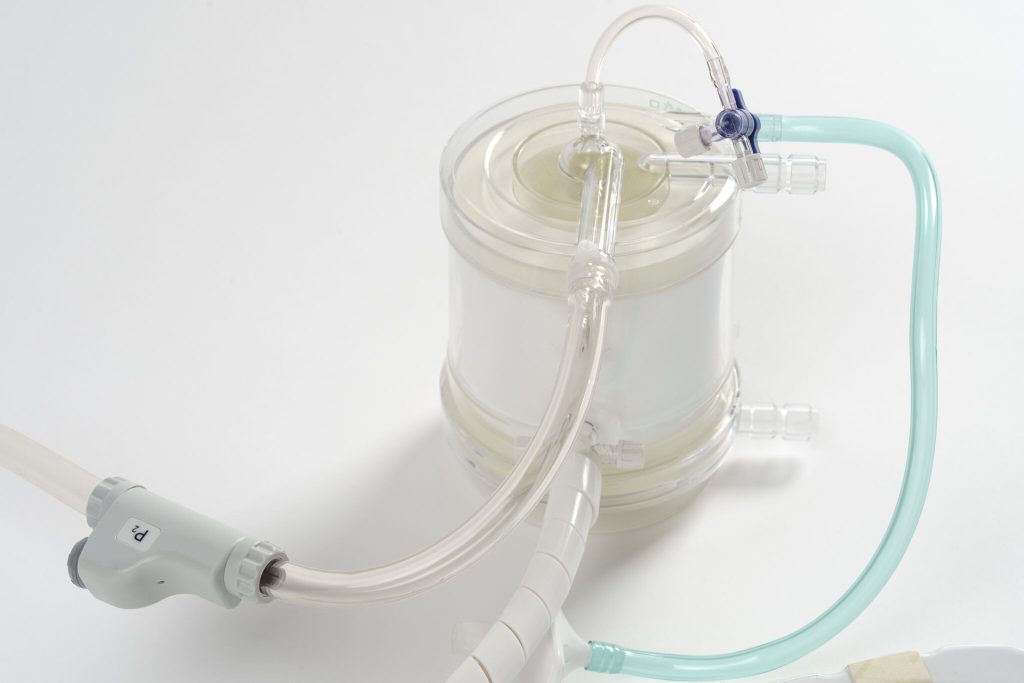
Detail of the oxygenator and connections of the Extra Corporeal Membrane Oxygenation circuit, with a layout designed for flow stability and operational continuity in long-term therapies, key elements for limiting unplanned interruptions and supporting the work of perfusionists in highly complex clinical settings (Photo: Lifemotion)
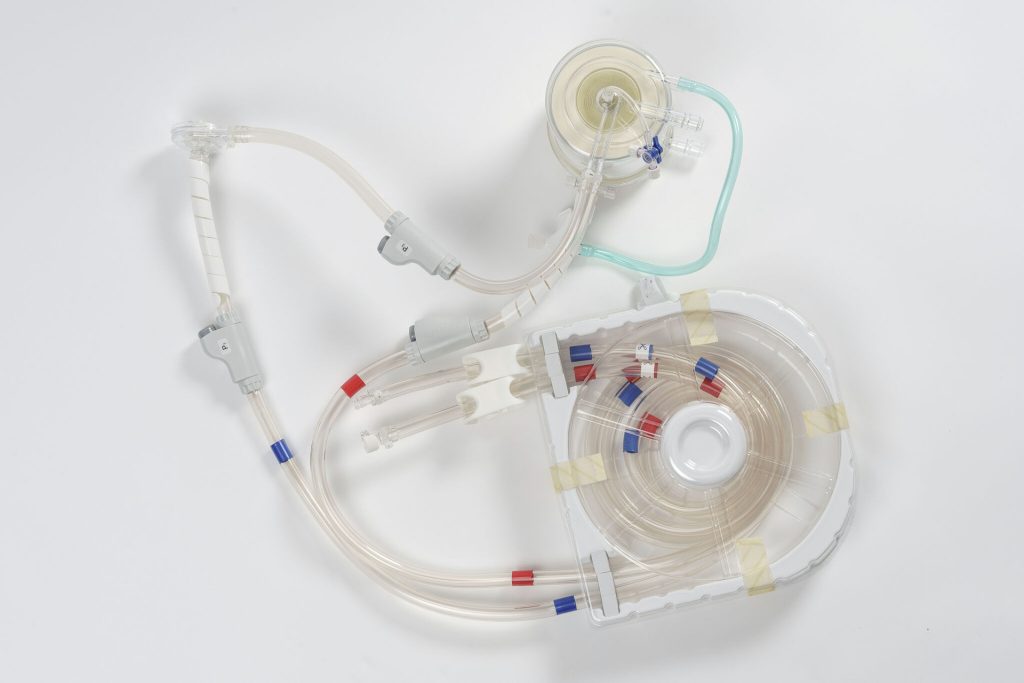
Top view of the Extra Corporeal Membrane Oxygenation circuit with arterial and venous lines organized to minimize space and risk of error during setup, an aspect that affects the safety of procedures and the integration of the system into the daily workflows of intensive care units (Photo: Lifemotion)
4 Mobile trolley configuration of the ECMO system, designed to promote continuity of extracorporeal support during patient transfer between departments or operating rooms, with particular attention to mechanical stability, cable management and accessibility of the main operating components (Photo: Lifemotion)

Lifemotion’s compact ECMO system integrates pump, oxygenator and circuit in an architecture designed to reduce priming volumes and start-up times, improving response speed in critical situations and promoting smoother management at the patient’s bedside in intensive care units and during intrahospital transport phases (Photo: Lifemotion)

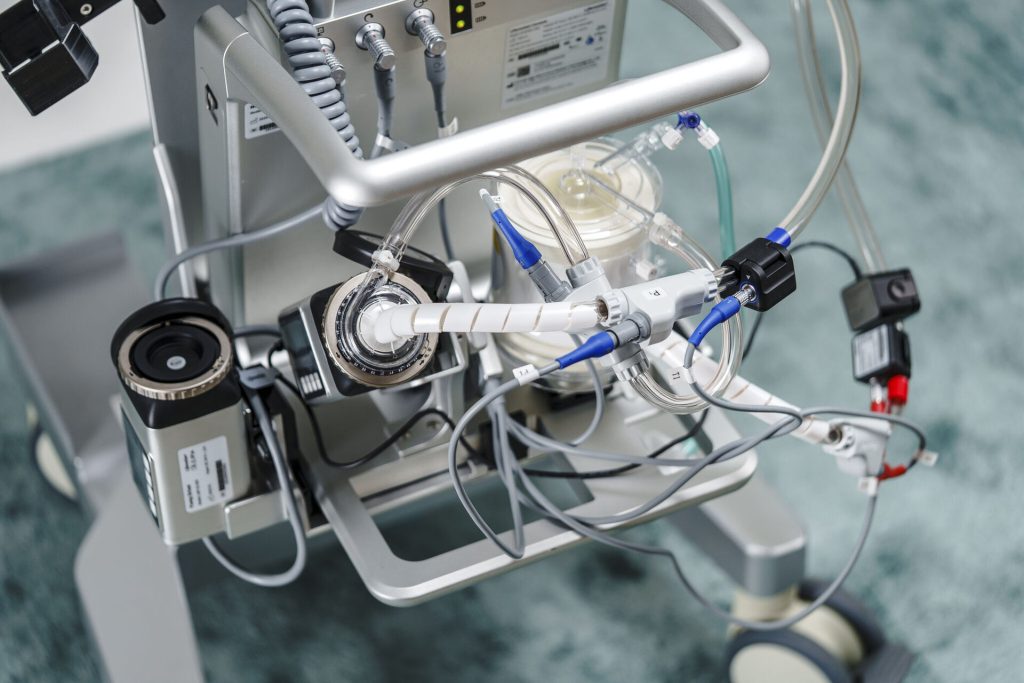
Air-oxygen mixing and flow control module, part of the technological ecosystem that allows fine-tuning of respiratory parameters in the ECMO circuit, an essential element for adapting extracorporeal support to the evolving clinical conditions of the patient in intensive care (Photo: Lifemotion)
Global expansion requires adapting to different healthcare systems. What lessons have you learned from entering new markets like Europe, Latin America, and the United States?
It’s crucial for us to continue following the mantra created by our CEO, Ms. Yang Liu, which is ‘Create Value for All,’ meaning creating value in all patient care and long-term support. We want to make the technology accessible to all markets, both developed and developing, and make it proportional to what local healthcare systems can afford. This is our CEO’s vision for ‘Creating Value,’ without expecting the cost of the treatment to be the same in every country. We set prices based on the market, respecting healthcare reimbursement costs and recognizing that access to this therapy shouldn’t be limited to developed countries alone.
Education and training are key elements for the adoption of critical technologies. What role do tools like augmented reality, virtual reality, and immersive simulations play in your educational programs?
We are currently very focused on the global launch of the product and its subsequent commercialization. We are training our sales channels to be as competent and professional as the Lifemotion International team. Furthermore, in the coming years, we will continue our work with ELSO internationally and with key centers in various countries. As we consolidate our presence in Europe, the Americas, Africa, and the Middle East, it will also be crucial to support the ELSO organization globally in its efforts to standardize patient care for this highly complex therapy.
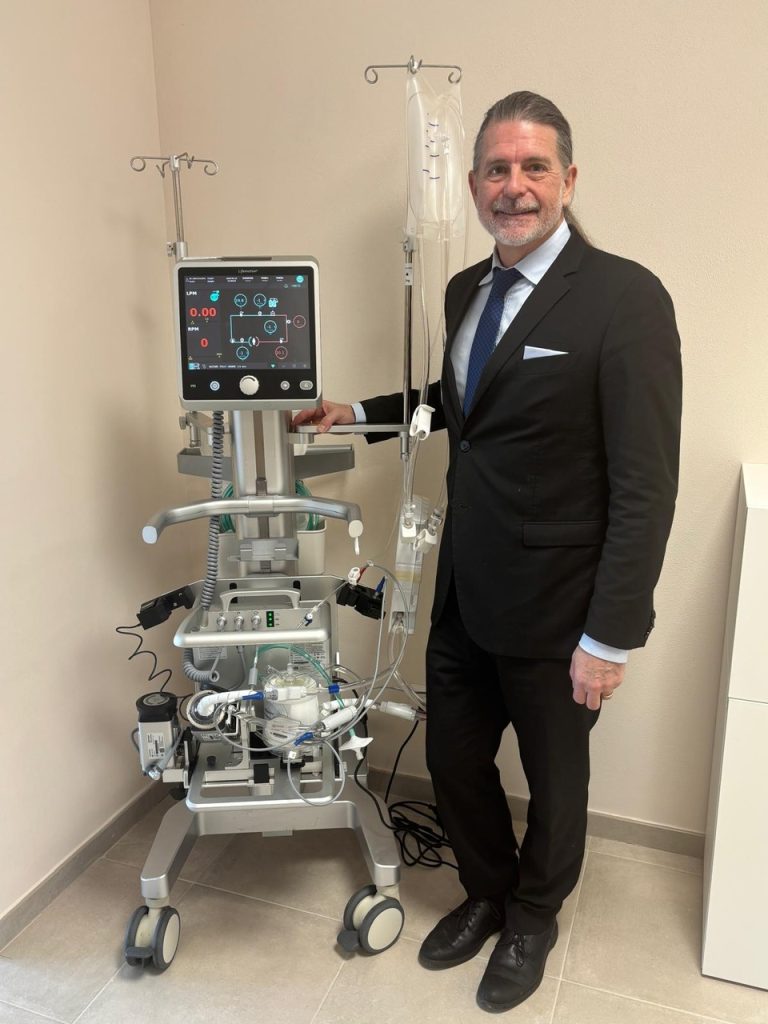
The founder of Lifemotion in Mirandola stands next to the complete ECMO system, with the circuit mounted on the mobile cart, underlining the direct relationship between entrepreneurial vision and the engineering of the device, designed to integrate into the workflows of intensive care units and patient transport procedures (Photo: Alice Luppi/Lifemotion)

Close-up detail of the facade of the Lifemotion headquarters in Mirandola, with the company logo integrated into the building’s architecture, representing the company’s local roots and its relationship with the local manufacturing sector specializing in intensive care technologies (Photo: Alice Luppi/Lifemotion)
You’ve often cited the Mirandola biomedical district as a key milestone in your professional career. What role does the Italian ecosystem play today in your innovation and growth strategy?
I’ve been fortunate to learn from the best experts in the field during my twenty-five years here in Mirandola. I arrived with very different ideas than those in the district about what is possible and what is not. I’ve often been told, ‘We’ve tried it, and it’s not practical,’ but over time, those same ideas have been put into practice in clean rooms and research and development laboratories. We need to redefine what is possible and what is not, and realize that nothing is impracticable or impossible if we work well and as a team. This mindset is crucial, as we must continue to address the challenges that present obstacles to this therapy, including costs and the high level of training required to make it work. With our mindset and work ethic, not only here in Biomedical Valley, but also in Shenzhen, I believe it’s possible to achieve our vision: to make this therapy safer, simpler, and with far fewer complications for patients.
What recent clinical findings or use cases can you share that concretely illustrate the impact of the Lifemotion system on patients’ lives and healthcare team organizations?
“What continues to be a pleasant surprise is how easily our system is portable. The modularity really lends itself to ease of use and transport within and outside the hospital. When I see messages reaching our clinical team about transport under extreme conditions in South America, Germany, or South Africa, it’s deeply moving. I realize how much trust our clinical partners have in us and our technology, because every successful treatment saves a life. These individual stories are incredibly inspiring and drive us all forward as we strive to push the limits of what is truly possible.”
“Anchor Entrepreneurship and Industry Catalysis”: the history of the Emilian biomedical district
From earthquake to reconstruction: the complete rebirth of Mirandola’s biomedical district
Mario Veronesi tells Confindustria about the birth of the Italian Silicon Valley in Mirandola
Here are three insights that might interest you:
Bonsai geopolitics and global districts: the Mirandola case conquers
Mario Veronesi, already seven years without the knowledge of the great innovator
The key innovation of the biomedical district of Mirandola

Michael Van Driel is pictured in his office in an informal pose, in front of the computer used to coordinate Lifemotion’s business and product development activities, in an image that conveys the daily governance of a company committed to innovation in the ECMO sector (Photo: Alice Luppi/Lifemotion)



